
There can be small differences between the output of one electrode and another, as well as changes in the output of electrodes over time. They are used to calibrate the pH measurement system (electrode and meter). With automatic temperature compensation (ATC), the signal from a separate temperature probe is fed into the pH meter, so that it can accurately determine pH value of the sample at that temperature.īuffers are solutions that have constant pH values and the ability to resist changes in that pH level.
#Ph meter manual
With manual compensation, a separate temperature measurement is required, and the pH meter manual compensation control can be set with the approximate temperature value. The temperature compensation may be either manual or automatic. Temperature compensation is contained within the instrument, because pH electrodes and measurements are temperature sensitive. In fact, the thickness of the glass determines its resistance and affects its output. Not a terribly advanced nor “high tech” process but a very critical and important step in the electrode manufacturing. The special glass body of the electrode is blown to its configuration by glass blowers. With all the technological advancements of the last 30 to 40 years, pH electrode manufacturing remains an art. PH electrode technology has not changed much in the past 50 to 60 years.

In some cases, voltages can also be read for special applications or for use with ion-selective or Oxidation-Reduction Potential (ORP) electrodes. The pH meter is basically a high impedance amplifier that accurately measures the minute electrode voltages and displays the results directly in pH units on either an analog or digital display. The input impedance of the pH meter and leakage resistances are therefore important factors. The pH electrode has very high internal resistance, making the voltage change with pH difficult to measure.
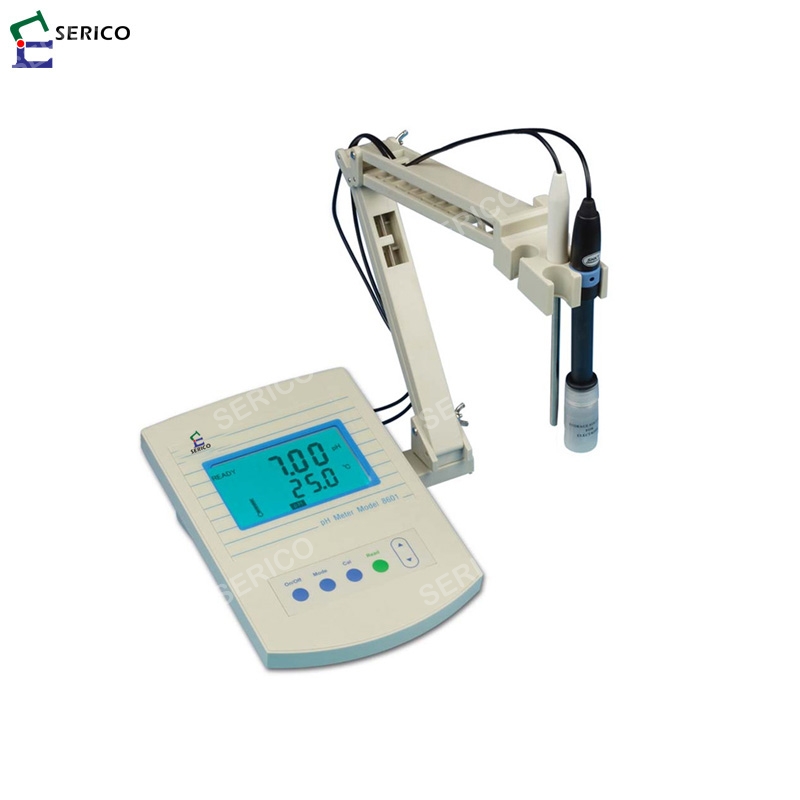
The reference electrode output does not vary with the activity of the hydrogen ion. The pH measuring electrode is a hydrogen ion sensitive glass bulb, with a millivolt output that varies with the changes in the relative hydrogen ion concentration inside and outside of the bulb. The pH electrode can be thought of as a battery, with a voltage that varies with the pH of the measured solution. A pH measurement system consists of three parts: a pH measuring electrode, a reference electrode, and a high input impedance meter. More accurate pH measurements are obtained with a pH meter. The term pH is derived from “p,” the mathematical symbol for negative logarithm, and “H,” the chemical symbol for Hydrogen. These indicators have limitations on their accuracy, and can be difficult to interpret correctly in colored or murky samples. The relationship between hydrogen ions and hydroxyl ions in a given solution is constant for a given set of conditions, either one can be determined by knowing the other.Ī rough indication of pH can be obtained using pH papers or indicators, which change color as the pH level varies.
#Ph meter free
Acids and bases have free hydrogen and hydroxyl ions, respectively. If equal amounts of H+ and OH- ions are present, the material is neutral, with a pH of 7. If the OH- concentration is greater than H+, the material is basic, with a pH value greater than 7. If the H+ concentration is greater than OH-, the material is acidic i.e., the pH value is less than 7. The pH value of a substance is directly related to the ratio of the hydrogen ion and the hydroxyl ion concentrations. The quantitative information provided by the pH value expresses the degree of the activity of an acid or base in terms of hydrogen ion activity. pH is the unit of measure that describes the degree of acidity or alkalinity. Contact an ATAGO representative for details.PH meter is an instrument used to measure acidity or alkalinity of a solution - also know as pH.

This may increase the lead time of the order by up to 2 - 3 weeks.

